Avera to enroll world’s first patients in new lung cancer trial
Jan. 19, 2018
By 2025, the way cancer is detected and treated will fundamentally change human health, predicted Avera’s Dr. Brian Leyland-Jones, in announcing the health system’s involved in a new worldwide lung cancer clinical trial.
“By 2025 this whole conundrum will be solved,” he said. “I truly believe the cancer paradigm will be solved. Metastatic cancer will disapper. Cancer will be picked up on a routine … exam”
That progress will be accelerated by efforts such as Avera is leading through the Worldwide Innovative Networking Consortium, he said. Avera will be the first in the world to enroll lung cancer patients in a clinical trial that is part of the international collaboration.
“With this trial, we don’t want to make an incremental improvement to lung cancer treatment outcomes. Instead we’re taking a bold step in a new direction so lung cancer patients have more hope after diagnosis,” said Dr. Benjamin Solomon, the lead investigator for the study at Avera and medical oncologist with Avera Cancer Institute.
“If successful, this study could lead to a complete paradigm shift in our approach to lung cancer treatment with a goal of realizing big improvements in outcomes for patients.”
More than 60 percent of non-small-cell lung cancer cases are detected in an advanced stage, and less than 5 percent of these patients are alive five years after diagnosis.
In the Survival Prolongation by Rationale Innovative Genomics, or SPRING, trial, patients will be given a three-drug protocol that incorporates immunotherapy called avelumab and two other targeted therapies called palbociclib and axitinib. All patients will be given the same drug combination, and their response rates will be tracked to see which patients respond best.
Through the WIN Consortium, an Avera research team led by Solomon helped design and implement the clinical trial from the ground up.
“This breakthrough research is happening in Sioux Falls, S.D., because of the high level of expertise and commitment available here,” said Dr. Vladimir Lazar, founder and chief scientific and operating officer of the WIN Consortium. “With these clinical trials, we want to go beyond what is now possible so patients around the world can have more hope when they get a lung cancer diagnosis.”
WIN “was created to push beyond what is done today in standard of care. It’s a huge effort and involves 40 centers in 19 countries,” he explained. “The beginning started in Sioux Falls and not elsewhere in the world. And that means something. That means here we have one of the most committed teams in the world.”
Avera’s commitment and passion for patients “is unique,” he added. “I haven’t seen that in any WIN consortium member across 19 countries.”
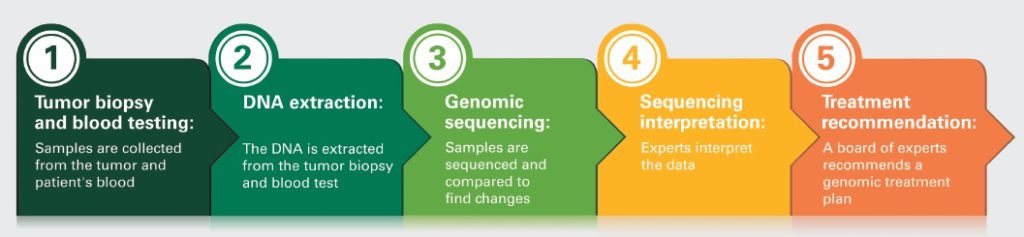
Because cancer evolves as it grows, it can acquire more genomic complexity over time. DNA sequencing and RNA expression levels in tumor and normal tissues will help guide care by analyzing the patient’s cancer genomic abnormalities to determine the specific changes that have occurred. A multidrug combination has the potential to treat lung cancer by blocking multiple cancer pathways that develop as a result of these genomic changes.
For the most effective results, researchers work to pair the drug combinations that may work best to combat each lung cancer genomic abnormality. To accomplish this, WIN developed an algorithm called SIMS, or Simplified Interventional Mapping System, which is hypothesized to better predict personalized treatment for cancer patients.
The end goal is to pair every lung cancer genomic abnormality with an effective drug combination. This may lead to dozens of clinical trials aimed at accomplishing this goal. In the future, this treatment algorithm potentially could be expanded to other tumor types, such as colon or breast cancer.
The SPRING trial will be led by Dr. Razelle Kurzrock of the University of California San Diego, Moores Cancer Center and Dr. Enriqueta Felip of the Vall d’Hebron Institute of Oncology in Spain. It will be launched in five countries and eight WIN member sites. Avera, one of two sites in the U.S., is the first to begin enrolling patients.
The SPRING trial will be conducted in two phases. Phase I will explore the safety of the drug combination and determine the optimal doses for Phase II, which will explore the efficacy of this tri-therapy regimen in first-line treatment of metastatic non-small-cell lung cancer. The trial will have strict eligibility criteria and limited enrollment.
“We hope the outcome of these studies will revolutionize patient care,” Solomon said. “We’re here to celebrate hope for more treatment options … This is the latest step in our fight against cancer: A fight we intend to win.”